DanDi Bioscience
- HOME
-
ABOUT US
-
WORK
-
PR
-
CAREERS
-
CONTACT US
-
ABOUT US
-
WORK
-
PR
-
CONTACT US
-
RECRUITING
- HOME
-
ABOUT US
-
WORK
-
PR
-
CONTACT US
-
RECRUITING
HLB Science: Introduction to innovative technologies.
Sepsis Therapeutics
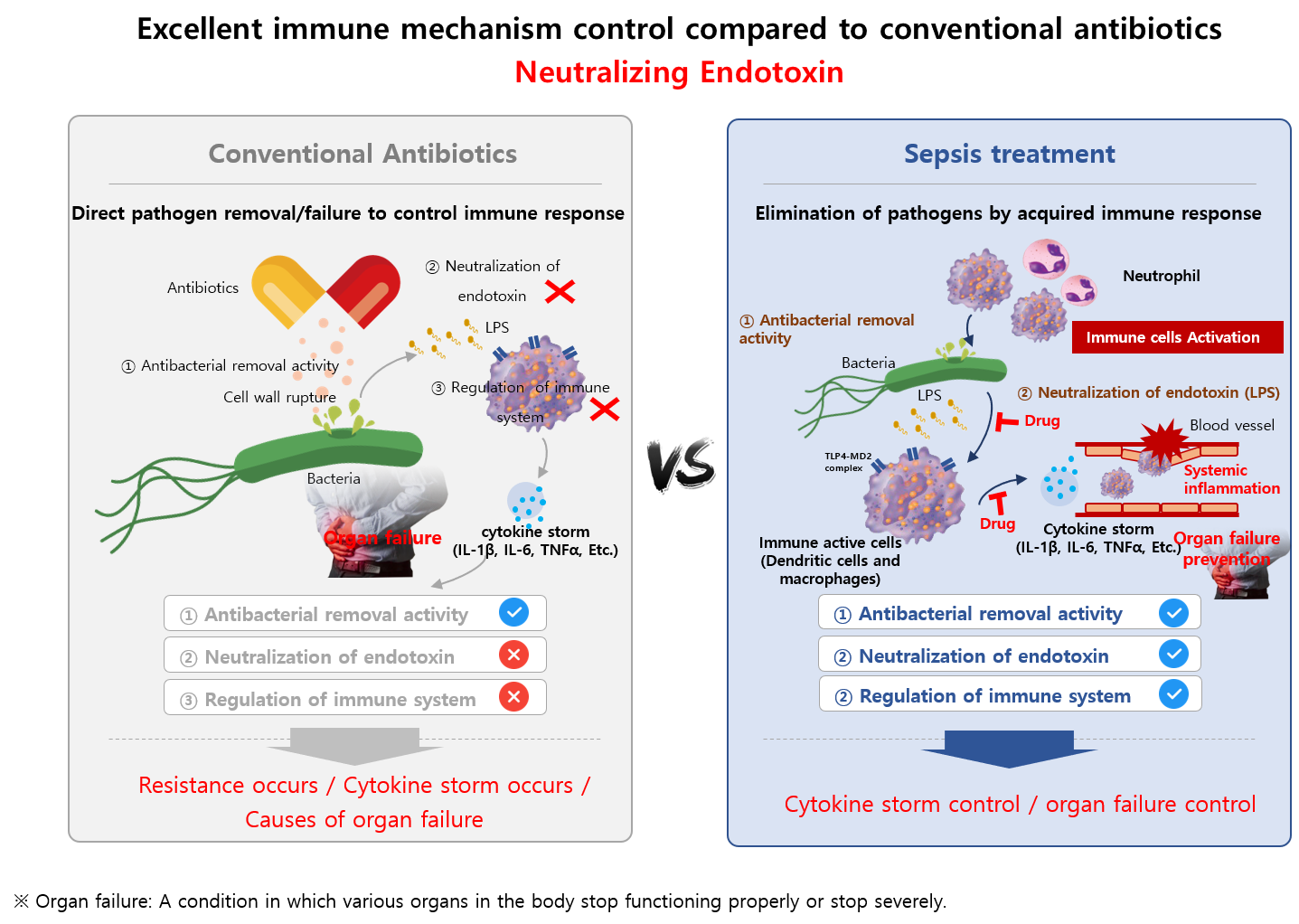
In order to control the secondary immune response initiated by the endotoxins released from gram-negative
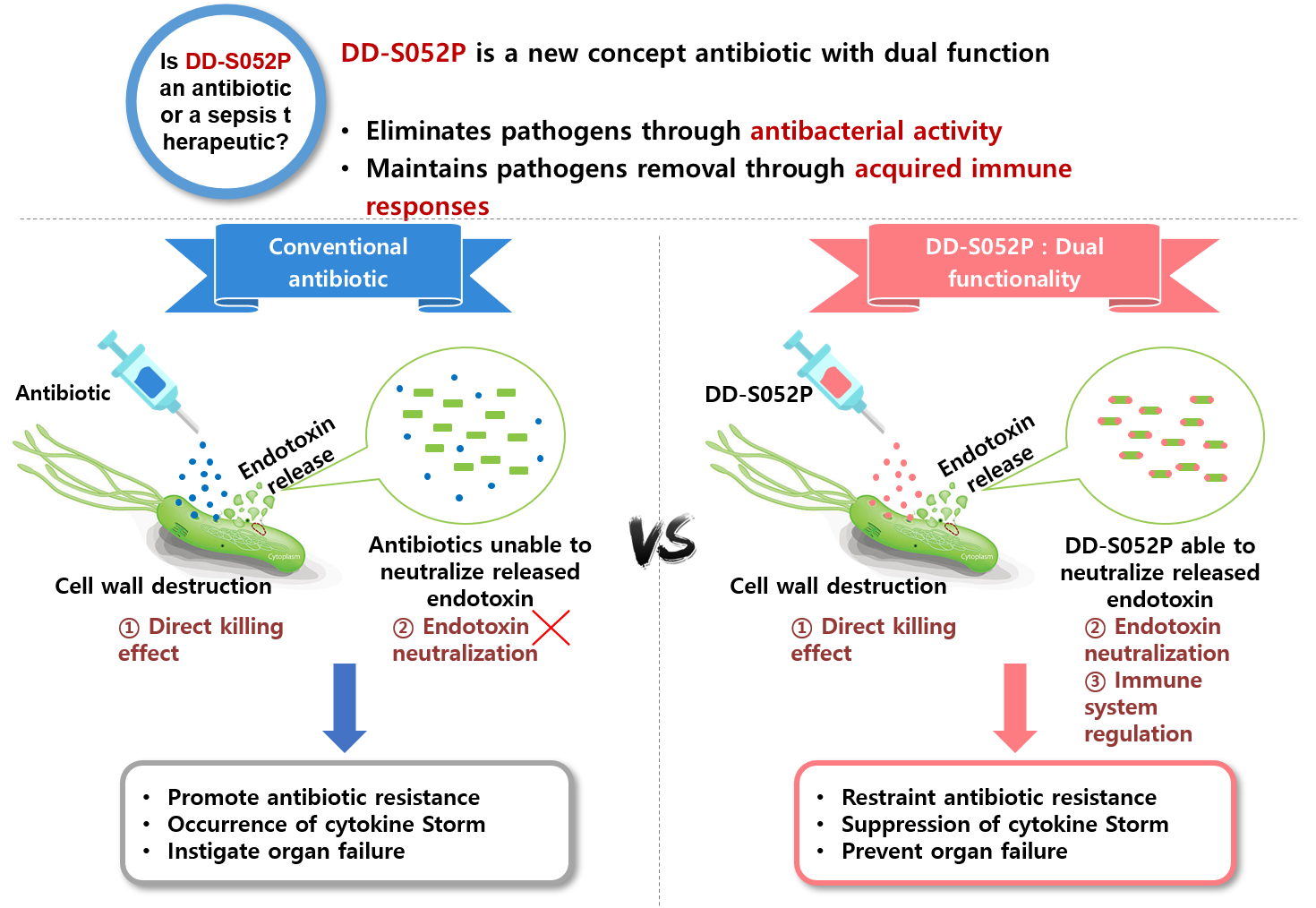
bacteria during sepsis treatment by antibiotics, our research institute has recently discovered a unique candidate “DD-S052P”.
Similar to polymyxin B (limited in clinical use due to the toxicity), “DD-S052P” has dual functions,
it directly kill the bacteria and binds the endotoxins released as a byproduct of the killing process.
With low toxicity, “DD-S052P” is a promising candidate to treat endotoxin releasing gram negative bacterial
infections and multidrug-resistant bacterial infections, which both are currently regarded as a serious public health issues.
Alzheimer's Disease Cure
An effective treatment strategy targeting Gram-negative bacteria and endotoxins that directly affect the onset of Alzheimer’s disease
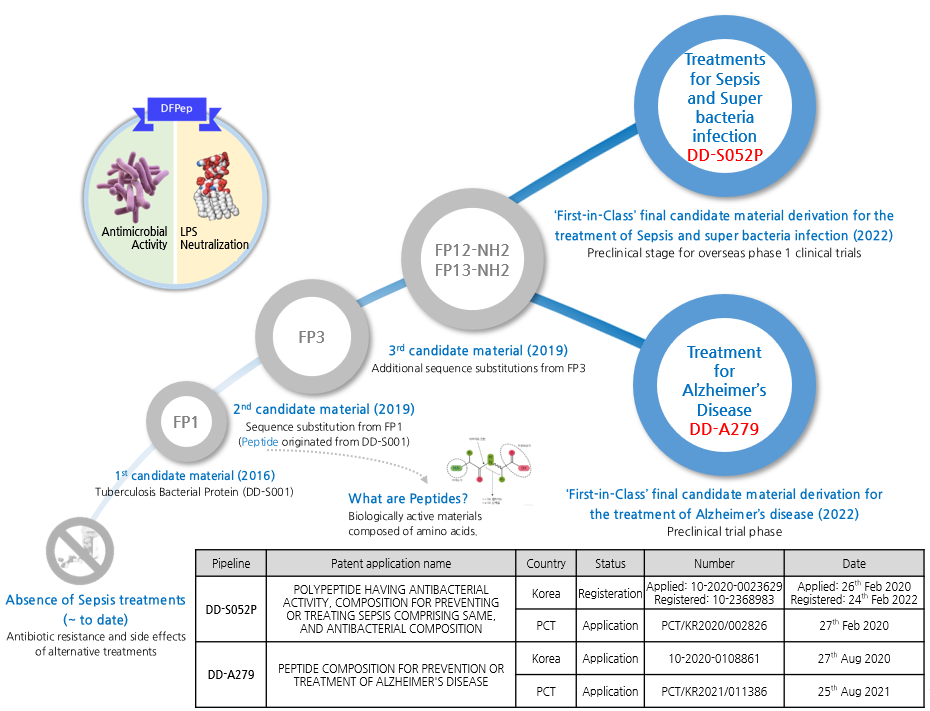
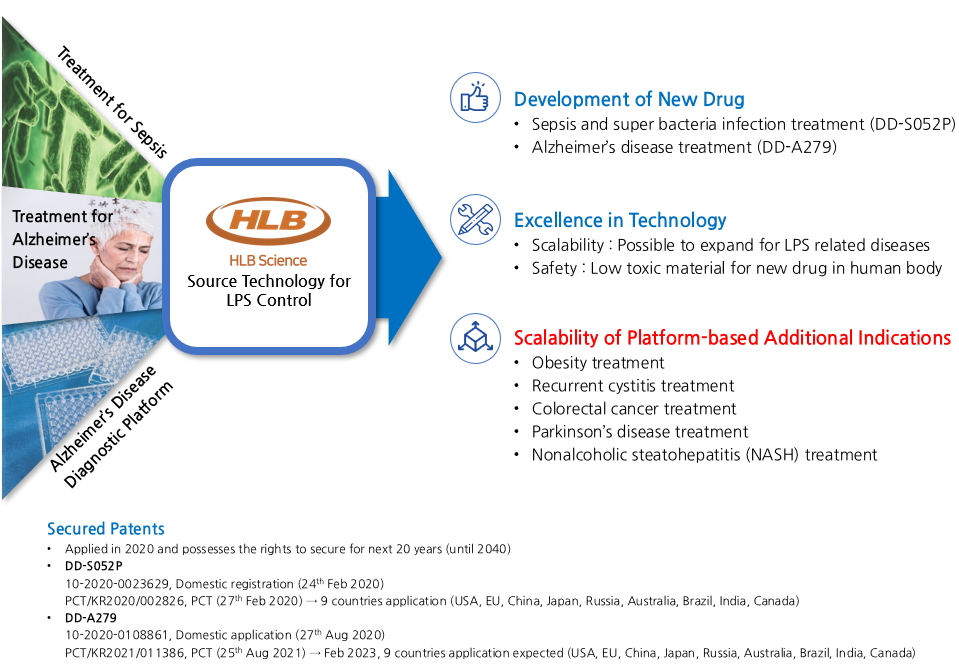
Development History and Scalability using DFPep-based